Key Takeaways:
Peptides are short amino acid chains that act as targeted signaling molecules.
Peptides like (BPC-157, TB-500, PT-141, Ipamorelin, Tesamorelin, GHK-Cu) have different mechanisms and uneven evidence.
BPC-157 & TB-500 → tissue repair → animal data, minimal human trials
Ipamorelin & Tesamorelin → growth hormone axis → limited vs strong clinical evidence
PT-141 → brain pathways → FDA-approved, notable side effects
GHK-Cu → repair signal → strong topical data, limited systemic evidence
Core pattern: → real biological effects, limited clinical validation
Core limitations: → injection required → rapid breakdown
We have all come across peptides in one way or another recently, whether that be on social media, in the news, or that friend who swears by peptides. But the overwhelming conversation has been around GLP-1 receptor agonists, the class of drugs that includes semaglutide. You know Ozempic, etc. This is understandable. GLP-1 drugs are the most commercially successful peptide therapeutics in history. Celebrities are endorsing it, influencers are talking about it. What this has done is get people talking about peptides like never before.
But there is a new crop of peptides that’s creating all the buzz in forums, clinics, and pharmacies. They do not belong to the same therapeutic class as semaglutides. BPC-157, TB-500, PT-141, Ipamorelin, Tesamorelin, and GHK-Cu are not metabolic drugs. They do not work through the same mechanisms. They do not share the same evidence base. More importantly, they do not fall under the same regulatory guidelines.
We at ONPEPS decided to take a closer and deeper look at these peptides and what they actually are. How they interact with the body and where the pharmaceutical science on each of them currently stands. Like you, we came across thousands of claims about these peptides in the wellness culture. So, we went through research and real-world evidence to get the facts right.
What Peptides Do That Other Drugs Can't
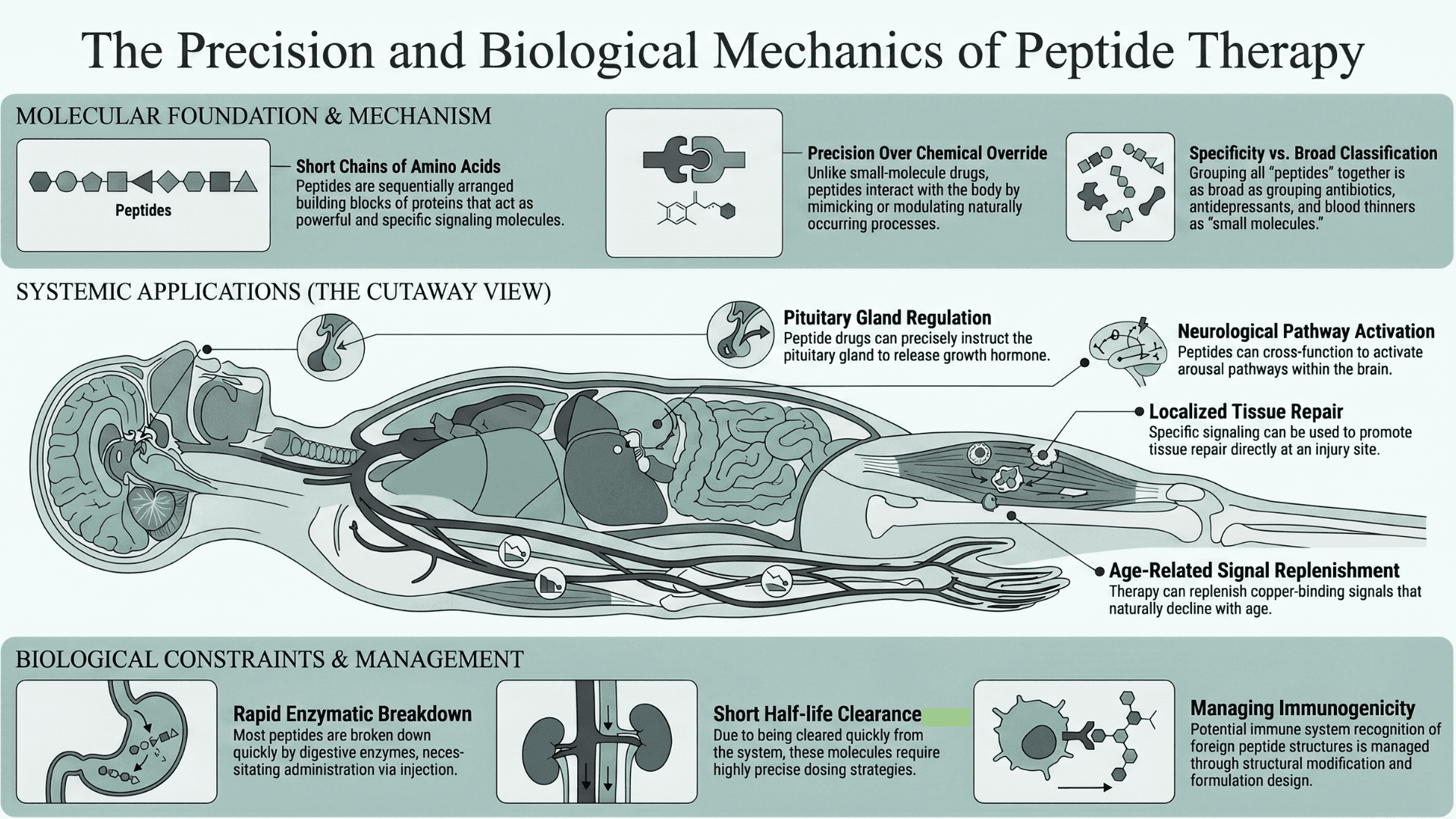
Peptides, as we already know, are short chains of amino acids, the same building blocks that make up proteins. They are sequentially arranged to make them powerful and specific signaling molecules. Their defining advantage over small-molecule drugs is precision. They interact with the body in ways that mimic or modulate naturally occurring processes.
A peptide drug can instruct the pituitary to release growth hormone or promote tissue repair at an injury site. It can also activate arousal pathways in the brain, or replenish a copper-binding signal that declines with age. These are distinct mechanisms operating in different parts of the human body.
So while grouping them together under the label peptides is accurate, it is not particularly illuminating. It’s the same as grouping antibiotics, antidepressants, and blood thinners as small molecules.
Peptides do, however, have a common biological weakness. Most are broken down rapidly by digestive enzymes, which is why nearly all peptide drugs require injection. Their short half-lives mean they are cleared quickly, requiring precise dosing strategies.
And you have to remember that immune systems can recognize foreign peptide structures in the body. This can cause immunogenicity. But it can be managed through structural modification and formulation design. For more information, take a look at our article on "How Therapeutic Peptides are designed and the future of Medicine".
So with all of these shared constraints, where does the science on each of these major peptides stand?
Tissue Repair or Recovery Peptides: BPC-157 and TB-500
BPC-157: The Wolverine Peptide & the Evidence Behind the Hype
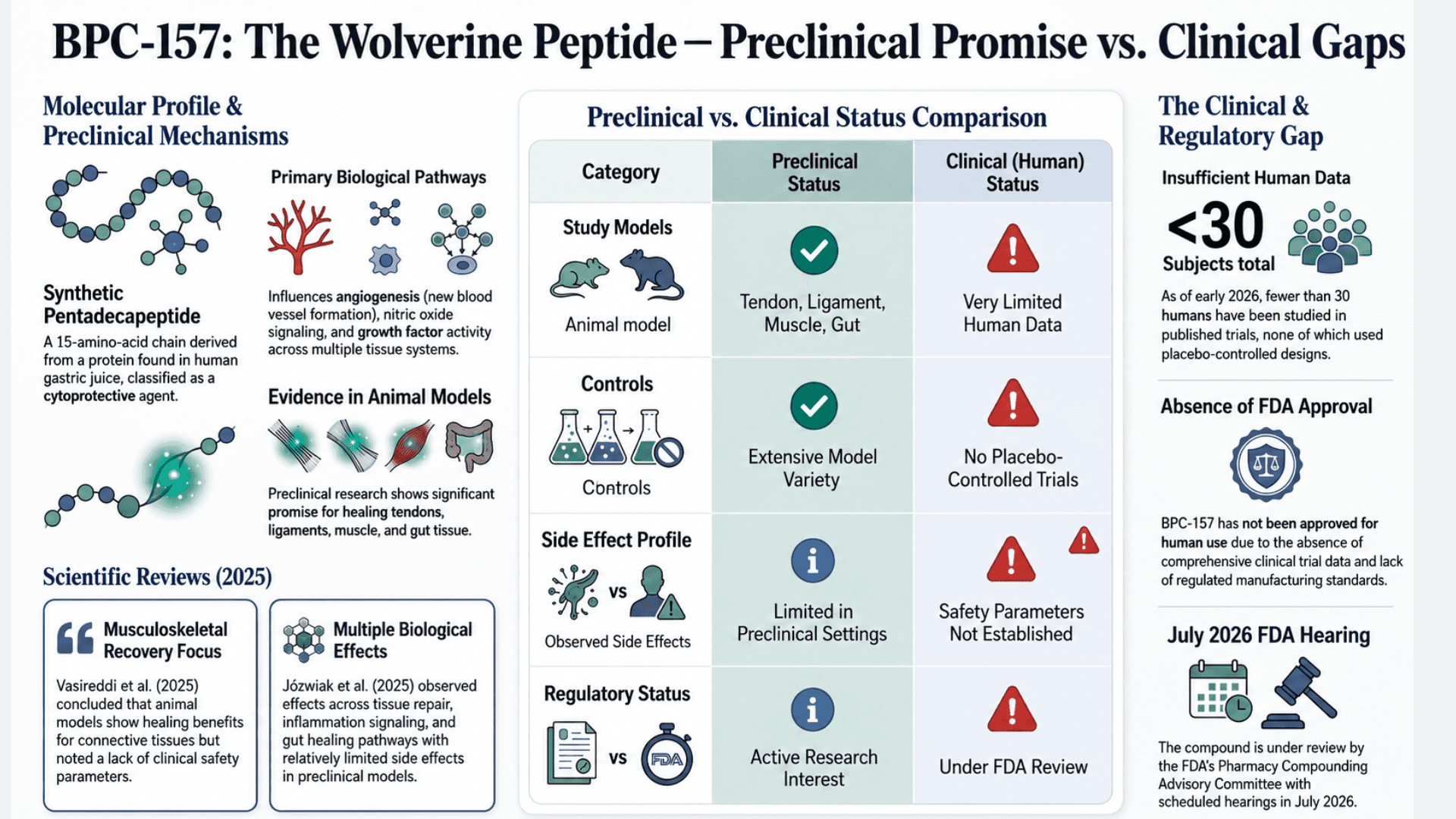
BPC-157 (Body Protection Compound-157) is often referred to online as the Wolverine peptide. It has become one of the most discussed recovery peptides in fitness and biohacking communities. Scientifically, it is a synthetic pentadecapeptide, a 15-amino-acid chain. It is derived from a protein found in human gastric juice.
In drug studies, it is classified as a cytoprotective agent. Preclinical research shows effects on angiogenesis (the formation of new blood vessels), nitric oxide signaling, and growth factor activity across multiple tissue systems, including tendon, ligament, muscle, and gut.
A 2025 systematic review published in Orthopaedic Sports Medicine (Vasireddi et al.) reviewed the full body of research surrounding BPC-157 and musculoskeletal injury recovery. The authors concluded that the peptide shows promise in animal models involving tendon, ligament, muscle, and connective tissue healing.
But they also found that adverse effects are possible because manufacturing standards are not regulated. There is also an absence of clinical safety parameters in humans. This isn't simply scientific caution. It reflects the current status of scientific evidence.
A separate 2025 literature and patent review published in Pharmaceuticals (Józwiak et al.) reached a similar conclusion. The review found that BPC-157 has demonstrated pleiotropic beneficial effects across multiple preclinical models. It also appears to maintain a relatively limited side effect profile within those settings. In other words, researchers observed effects across multiple biological processes, including tissue repair, inflammation signaling, blood vessel formation, and gut healing pathways.
However, the compound has not been approved for human use by the FDA or equivalent global regulatory agencies. This is due to the absence of comprehensive clinical trial data. As of early 2026, fewer than 30 humans have been studied across all published BPC-157 trials. None of those studies included placebo-controlled designs.
That gap between a compelling preclinical record and an absent clinical one is not evidence of failure. It only highlights a research timeline that has not yet reached the stage of human validation.
BPC-157 is currently under review by the FDA's Pharmacy Compounding Advisory Committee, with hearings scheduled for July 2026.
TB-500: Thymosin Beta-4, Peptide Stacking, and Recovery Claims
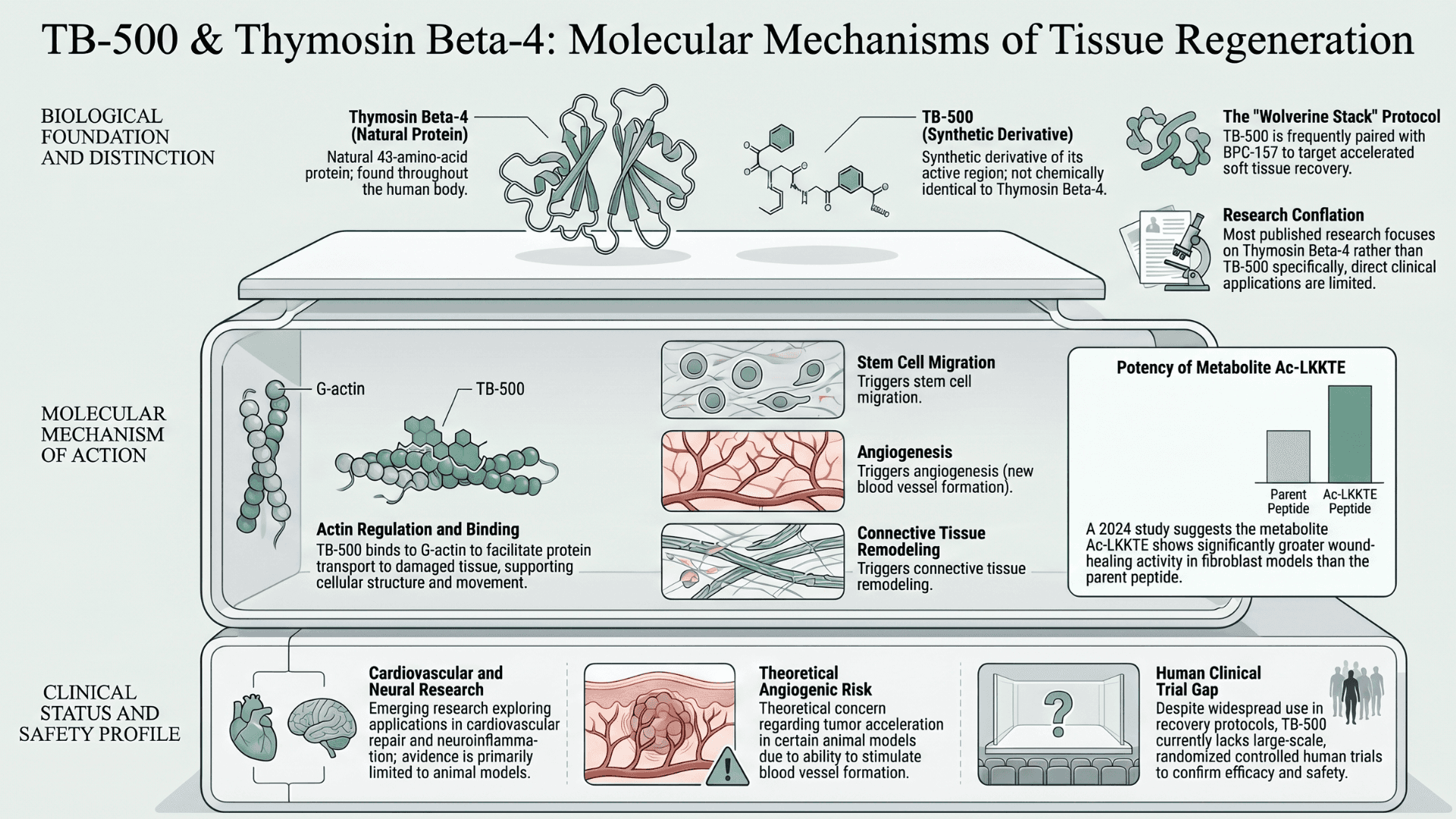
TB-500 is a synthetic research peptide derived from the active region of Thymosin Beta-4, a naturally occurring 43-amino-acid protein. It is found throughout the human body and is heavily involved in cellular movement, wound healing, and tissue regeneration (Maar et al.).
Online, it is frequently grouped with BPC-157 in what is known as the Wolverine stack recovery protocols. That is because real-world users have experienced accelerated healing and soft tissue recovery.
One of its key proposed mechanisms involves actin regulation. TB-500 binds to G-actin, a protein involved in cellular structure and movement, and helps transport it to areas of tissue damage where repair activity is taking place.
Researchers have also observed downstream effects involving stem cell migration, angiogenesis, inflammatory modulation, and connective tissue remodeling during healing (Maar et al.). Interest in Thymosin Beta-4 research has also expanded into areas involving cardiovascular repair, neuroinflammation, and tissue remodeling. Still, most of that evidence comes from laboratory and animal models rather than large-scale human trials.
Recent research has also suggested that some of the wound-healing activity historically attributed to TB-500 may depend more heavily on downstream metabolites. These are produced after the peptide is broken down in the body rather than the parent peptide itself. A 2024 study identified the metabolite Ac-LKKTE as showing significantly greater wound-healing activity in fibroblast models than TB-500 alone (Rahaman et al.).
One important distinction often lost in online discussions is that most of the published research focuses on Thymosin Beta-4 itself rather than TB-500 specifically.
While TB-500 is designed around the active region of the larger peptide, the two are not chemically identical, and findings surrounding one cannot automatically be applied to the other. That distinction matters. Many of the claims circulating online about TB-500 recovery protocols are taken from broader Thymosin Beta-4 research. Clinical evidence directly related to TB-500 is still lacking.
As a result, findings involving one compound cannot automatically be generalized to the other.
The evidence base surrounding Thymosin Beta-4 includes extensive preclinical research alongside limited early-stage human safety investigation. But like BPC-157, it still lacks large-scale randomized controlled human trials.
One concern repeatedly discussed in the safety literature is the theoretical risk of tumor acceleration observed in certain animal models due to angiogenic activity (the ability to stimulate the formation of new blood vessels). But this has not been conclusively demonstrated in humans.
TB-500 is also increasingly associated with peptide stacking protocols involving BPC-157 and other recovery peptides in fitness and biohacking communities. This is a growing trend in spite of the absence of controlled clinical trials evaluating those combinations in humans (Rahaman et al.).
Growth Hormone Peptides: Ipamorelin and Tesamorelin
Ipamorelin: The Growth Hormone Peptide Used for Recovery and Body Composition
Unlike recovery peptides such as BPC-157 and TB-500, ipamorelin and tesamorelin work through the body’s growth hormone system. They do not introduce synthetic HGH directly into the bloodstream. Instead, these peptides stimulate the pituitary gland to increase the body’s own release of growth hormone through receptor signaling pathways.
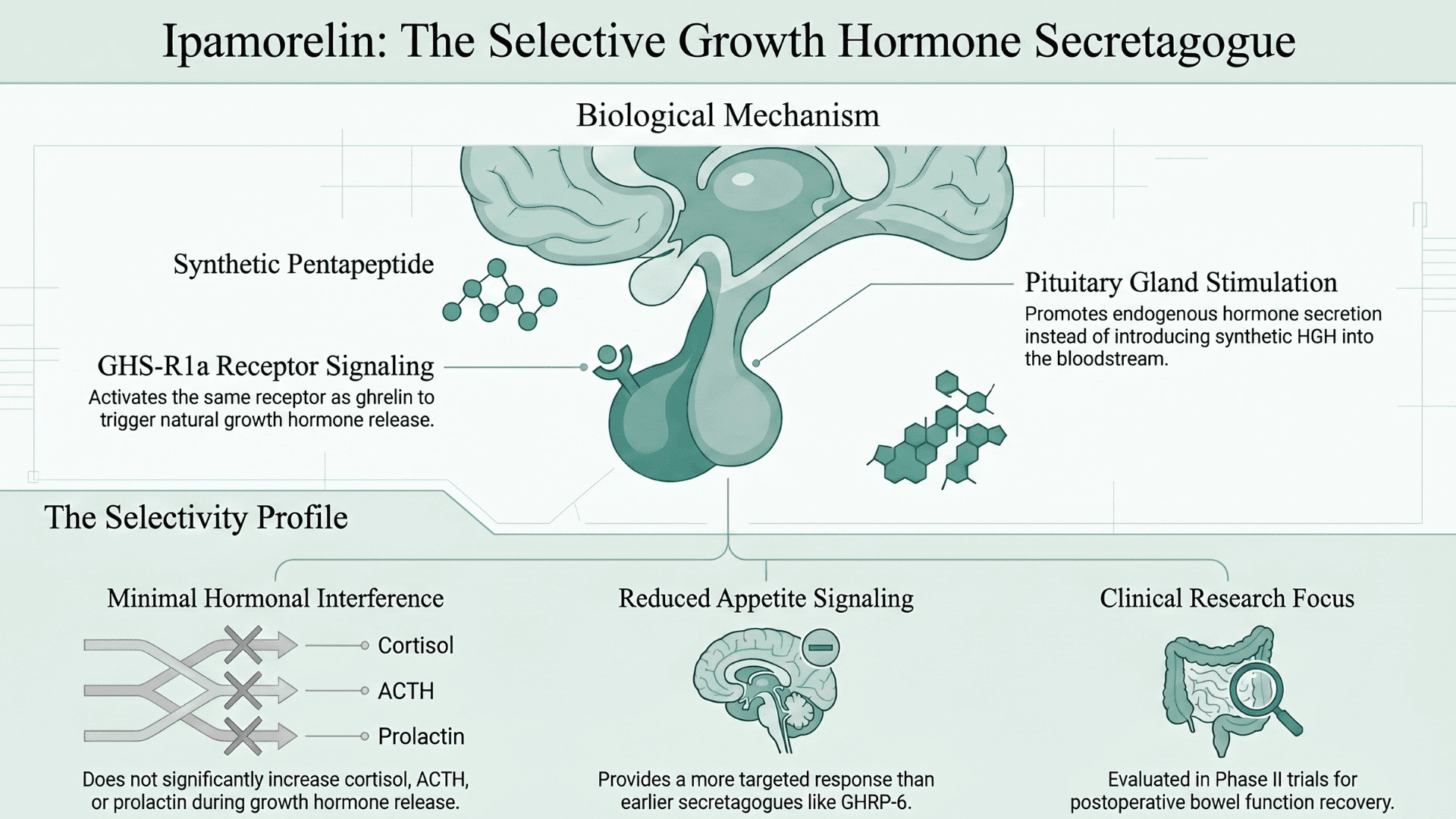
Ipamorelin is a synthetic pentapeptide and one of the first highly selective growth hormone secretagogues (a substance that stimulates the body to release more growth hormone). It was developed specifically to stimulate growth hormone release without significantly increasing cortisol, ACTH, prolactin, or appetite signaling.
Originally developed by Novo Nordisk and characterized in a landmark 1998 paper by Raun et al. in the European Journal of Endocrinology, ipamorelin acts through the growth hormone secretagogue receptor (GHS-R1a), the same receptor associated with ghrelin signaling. Ghrelin signaling refers to the biological signaling pathway activated by ghrelin, a hormone often called the 'hunger hormone'.
That selectivity profile is what made ipamorelin particularly attractive in anti-aging, recovery, and bodybuilding communities.
Earlier growth hormone secretagogues, such as GHRP-6, were associated with stronger appetite stimulation and broader hormonal effects. But ipamorelin was designed to produce a more targeted GH response.
Human studies have included trials in healthy volunteers to understand how ipamorelin behaves in the body. This was done alongside a Phase II randomized controlled trial involving 87 bowel surgery patients. The research investigated whether the peptide could help restore normal bowel movement after surgery (Beck et al.).
Despite ongoing interest in fitness and longevity circles, large-scale human trials evaluating ipamorelin's use for muscle growth, anti-aging, or athletic recovery remain limited.
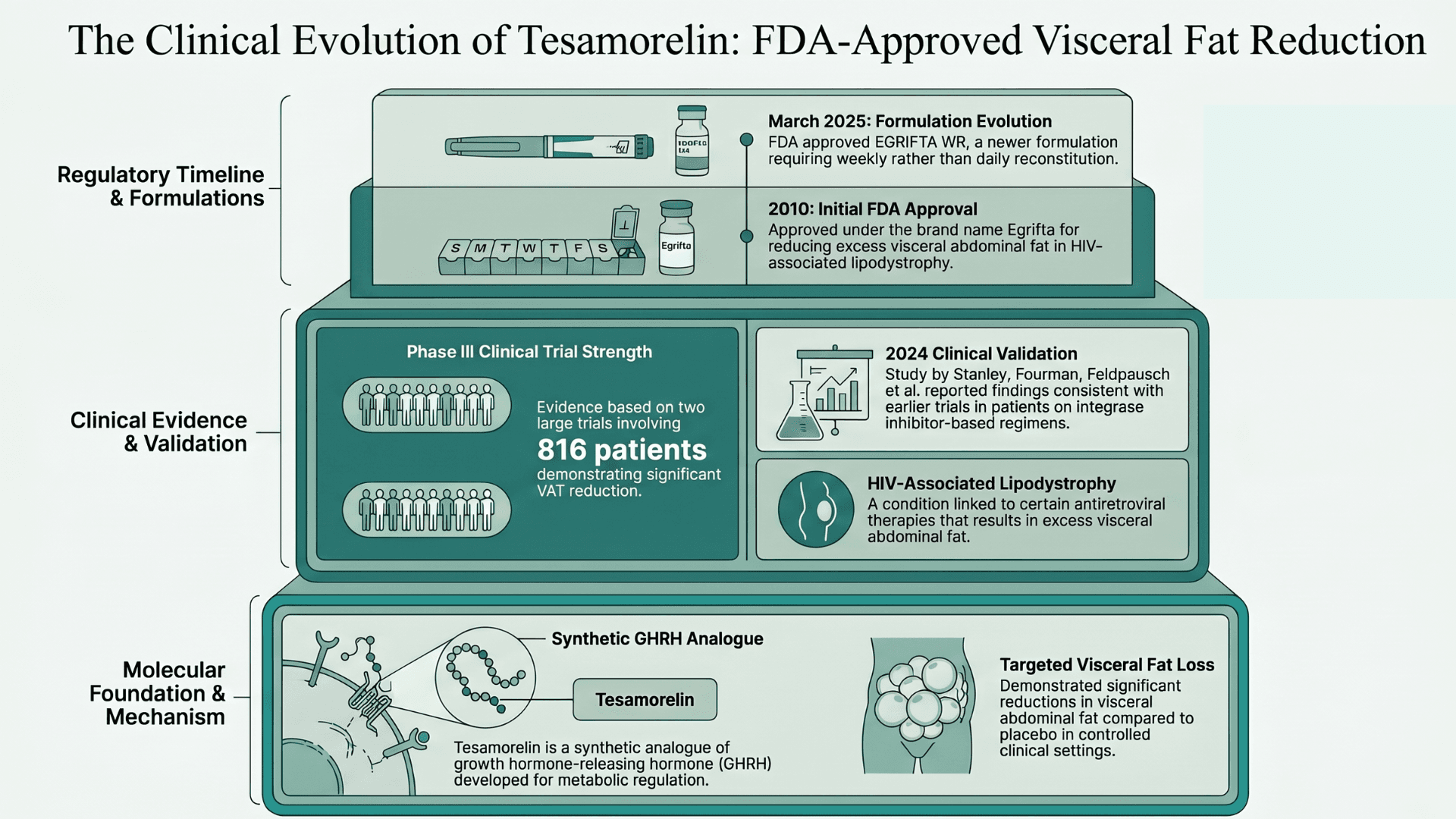
Tesamorelin: The FDA-Approved Peptide for Fat Loss
Tesamorelin occupies a very different position on the evidence spectrum because it is fully FDA-approved. It was first developed as a synthetic analogue of growth hormone-releasing hormone (GHRH).
Tesamorelin was approved in 2010 under the brand name Egrifta for reducing excess visceral abdominal fat in HIV-associated lipodystrophy. It is a condition linked to certain antiretroviral therapies.
In March 2025, the FDA approved a newer formulation, EGRIFTA WR, requiring weekly rather than daily reconstitution (FDA).
The clinical evidence behind tesamorelin is substantially stronger than most peptides discussed in online recovery and biohacking communities. Two large Phase III clinical trials involving 816 patients demonstrated significant reductions in visceral abdominal fat compared to placebo.
A 2024 study examining tesamorelin in HIV patients receiving integrase inhibitor-based regimens reported findings consistent with earlier clinical trials (Stanley, Fourman, Feldpausch et al., 2024).
Yes, it's true that tesamorelin is often discussed online in the context of general fat loss, longevity, and body composition. But the strongest clinical evidence remains tied specifically to HIV-associated lipodystrophy rather than broader off-label use.
PT-141: The Libido Peptide That Works Differently Than Viagra
PT-141 (bremelanotide, brand name Vyleesi) is a synthetic cyclic heptapeptide derived from alpha-melanocyte-stimulating hormone (α-MSH). Its mechanism is different from any other compound discussed here. PT-141 acts centrally through melanocortin pathways, rather than peripherally through vascular or tissue mechanisms.
Specifically, PT-141 activates melanocortin-3 and melanocortin-4 receptors (MC3R and MC4R) in hypothalamic and limbic regions. These areas of the brain control sexual motivation, arousal, and reward. This is why its effects on sexual function appear even in the absence of direct physical stimulation.
PT-141 also works in patients who have not responded to PDE-5 inhibitors such as sildenafil.
PT-141 carries FDA approval for premenopausal women with hypoactive sexual desire disorder (HSDD). This makes it one of the few compounds in this group with a completed regulatory pathway. The approval is based on data from multiple clinical trials.
In men, its use remains off-label, though the mechanism of action through shared melanocortin receptor pathways applies across sexes (FDA Vyleesi Prescribing Information).
Now we need to talk about the side effects, particularly given the increasing usage within the peptide community. Nausea occurs in up to 40% of subjects at standard doses, flushing in 20–40%, and transient blood pressure elevation is documented. Tolerance reduction with repeated use has also been noted.
We are not saying the compound is bad. Rather, it shows the difference between dosage and usage conditions in a clinical setting and in the real world when sourced from gray markets.
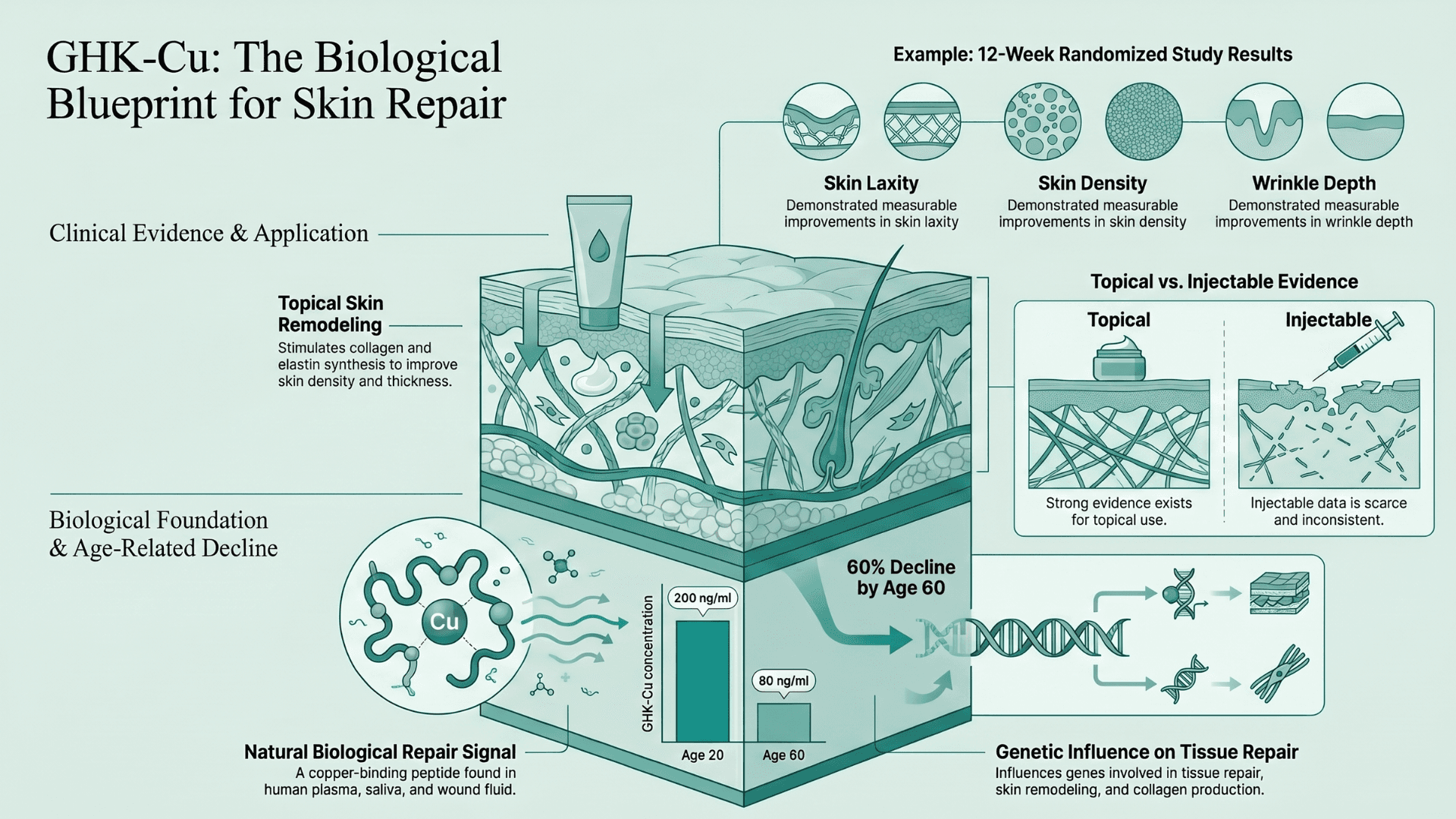
GHK-Cu: The Copper Peptide People Use for Skin Repair and Anti-Aging
GHK-Cu is a naturally occurring copper-binding peptide found in human plasma, saliva, and wound fluid. It is primarily studied for its role in skin repair, tissue regeneration, and aging research. It is released during tissue repair processes and functions as a biological repair signal in the body.
GHK-Cu levels naturally decline with age, dropping from roughly 200 ng/ml at age 20 to around 80 ng/ml by age 60. No wonder it is one of the most researched peptides when it comes to skin aging and tissue repair.
The clinical evidence for GHK-Cu is the one area where human trial data genuinely exists at scale. Multiple controlled human studies have demonstrated measurable effects on skin density and thickness, collagen synthesis, wrinkle depth, and skin elasticity.
A 12-week randomized study in 71 women found improvements in skin density, laxity, fine lines, and wrinkle depth with GHK-Cu facial cream versus placebo. A 2025 review published in PMC also highlighted GHK-Cu’s role in stimulating collagen and elastin production. It was also seen influencing genes involved in tissue repair and skin remodeling.
But this is where we need to be careful, because these studies are related to GHK-Cu topical application. The moment we start talking about injectable use, there are very few studies. And the results of these studies do not match those of the topical evidence.
The Peptide Evidence Gap That Matters
So these are the top trending peptides. Taken together, we can see one consistent pattern. Many show promising early research, clear biological effects, and growing interest in areas such as recovery, body composition, libido, and skin repair.
But for most of these peptides, the strongest evidence still comes from preclinical studies and limited human trials. There is a severe gap in the large-scale clinical data normally required for full regulatory approval.
This is to be expected. Peptides exist in the gap between promising preclinical drugs and approved compounds. But the pharmaceutical pipeline for tissue repair, GH secretagogues, and melanocortin agonists is active. Dozens of related compounds are in late-stage development or clinical evaluation.
So the main issue is that most peptide users are likely to take these compounds without proper clinical oversight or biomarker baselines. Moreover, gray-market sourced peptides often are not quality-controlled, so safety remains a concern. But we at ONPEPS will continue to dive into the latest developments so that you can make informed decisions when it comes to peptides.
FAQ:
What is the difference between lyophilized peptides and pre-mixed liquids?
Lyophilized (freeze-dried) peptides remain stable for years when frozen, whereas pre-mixed liquid formulations rapidly degrade and lose efficacy due to water exposure.Why must peptides be reconstituted with bacteriostatic water?
Bacteriostatic water contains 0.9% benzyl alcohol, which prevents bacterial growth and keeps the peptide sterile for multi-dose use over its 14 to 28-day refrigerated lifespan.Do peptide receptors downregulate, and do they require cycling?
Yes, continuous exposure can desensitize surface receptors, so most protocols incorporate "on" and "off" cycles to maintain biological response and prevent antibody buildup.How do peptides differ from SARMs or anabolic steroids?
Peptides bind to surface receptors (GPCRs) to stimulate natural bodily processes, whereas steroids enter the cell nucleus to alter gene expression and suppress endogenous hormone production.Why are CJC-1295 and Ipamorelin frequently stacked together?
They act synergistically. Ipamorelin triggers an immediate, acute pulse of natural growth hormone, while the extended half-life of CJC-1295 prolongs the baseline elevation of that pulse.Does Ipamorelin cause HPA axis suppression?
No, Ipamorelin is a highly selective secretagogue engineered to stimulate growth hormone release without significantly spiking the cortisol or prolactin levels that disrupt the HPA axis.What is the biological difference between TB-500 and Thymosin Beta-4?
Thymosin Beta-4 is a full-length, naturally occurring 43-amino-acid protein, while TB-500 is a synthetic fragment containing only the specific active region responsible for localized tissue repair.Are injectable GHK-Cu protocols different from topical application?
Yes, topical GHK-Cu is backed by robust human data for skin remodeling, while systemic injections lack clinical trials and carry theoretical risks of copper accumulation if dosed improperly.What does the FDA’s July 2026 PCAC review mean for BPC-157?
The July 2026 hearings will officially determine if BPC-157 and other Category 2 peptides can be legally produced by 503A compounding pharmacies, or if they will remain strictly banned.What is the legal difference between "RUO" labeling and a compounded prescription?
Compounded prescriptions are rigorously tested and regulated by licensed pharmacies, whereas "Research Use Only" (RUO) peptides bypass FDA oversight and lack guaranteed purity, sterility, or dosage verification.
Sources:
Józwiak, Bauer, Kamysz, Kleczkowska — “Multifunctionality and Possible Medical Application of the BPC 157 Peptide — Literature and Patent Review.” Pharmaceuticals, MDPI, January 2025.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11859134/Vasireddi, Hahamyan, Salata et al. — “Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review.” Orthopaedic Journal of Sports Medicine, SAGE, 2025.
https://journals.sagepub.com/doi/10.1177/15563316251355551Sikiric, Seiwerth, Skrtic et al. — “BPC 157 Therapy: Targeting Angiogenesis and Nitric Oxide's Cytotoxic and Damaging Actions.” Pharmaceuticals, MDPI, September 2025.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12567428/Rahaman, Muresan, Min et al. — “Simultaneous quantification of TB-500 and its metabolites in in-vitro experiments and rats by UHPLC-Q-Exactive orbitrap MS/MS.” Journal of Chromatography B, 2024.
https://pubmed.ncbi.nlm.nih.gov/38382158/Maar, Hetenyi, Maar et al. — “Utilizing Developmentally Essential Secreted Peptides Such as Thymosin Beta-4 to Remind the Adult Organs of Their Embryonic State.” Cells, 2021.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8226393/FDA Prescribing Information — Vyleesi (bremelanotide injection). Approved June 2019 for HSDD in premenopausal women.
https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/210557s000lbl.pdfPeptide Breakdown — “PT-141 (Bremelanotide): Research, Mechanism, and Clinical Evidence for Sexual Function.” Updated February 2026.
https://peptidebreakdown.com/peptides/pt-141/Raun, Hansen, Johansen et al. — “Ipamorelin, the first selective growth hormone secretagogue.” European Journal of Endocrinology, 1998.
https://pubmed.ncbi.nlm.nih.gov/9849822/Beck, Sweeney, McCarter — “Prospective, randomized, controlled, proof-of-concept study of the Ghrelin mimetic ipamorelin for the management of postoperative ileus in bowel resection patients.” Neurogastroenterology & Motility, 2014.
https://pubmed.ncbi.nlm.nih.gov/25331030/Ishida — “Growth hormone secretagogues: history, mechanism of action, and clinical development.” JCSM Rapid Communications, Wiley, 2020.
https://onlinelibrary.wiley.com/doi/full/10.1002/rco2.9FDA Prescribing Information — EGRIFTA WR (tesamorelin F8).
https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/022505s020lbl.pdfStanley, Fourman, Feldpausch et al. — “Efficacy and Safety of Tesamorelin in People with HIV on Integrase Inhibitors.” PMC, 2024.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11365754/Pickart, Vasquez-Soltero, Margolina — “The Human Tripeptide GHK-Cu in Prevention of Oxidative Stress and Degenerative Conditions of Aging.” Cosmetics, 2015.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6073405/Lupu, Graur et al. — “Peptides: Emerging Candidates for the Prevention and Treatment of Skin Senescence: A Review.” PMC, January 2025.https://pmc.ncbi.nlm.nih.gov/articles/PMC11762834/



