Peptide Fundamentals: Key Takeaways
Peptides are short chains of 2–50 amino acids linked by peptide bonds.
They are formed in cells through ribosomal synthesis or created in labs using chemical methods.
Their structure allows them to bind to receptors and regulate biological processes.
Peptides act as signaling molecules, hormones, immune defenders, and structural components.
They play key roles in metabolism, growth, and cellular communication.
Some peptides are used in medicine, especially for metabolic diseases and cancer.
Limitations include low oral absorption and rapid breakdown in the body.
Their function and impact depend on their sequence, structure, and chemical modifications.
What are Peptides made of?
Peptides are defined by research (Forbes Kaprive et al., NIH) as short strings of amino acids. They act as primary biochemical modulators for a wide range of biological activities in the human body.
Amino acids serve as the organic starting blocks. Every single amino acid possesses a carboxyl terminal and an amino group. These two components are the foundation of the entire structure.
The C-terminal is essentially the tail end of a peptide chain. Because proteins are built piece by piece, this is the very last amino acid added to the string.
It is much more than just a stopping point. It dictates where the peptide travels in the body, how long it lasts before breaking down, and how it actually does its job.
How are Peptides Formed?
Amino acid molecules connect together through a specific condensation reaction. This chemical process results in the permanent loss of a water molecule.
The remaining portion of each amino acid is called a residue. It is called a residue because it is the exact part left over after the dehydration reaction occurs. This reaction forms a peptide bond, which defines how stable and rigid the final structure becomes.
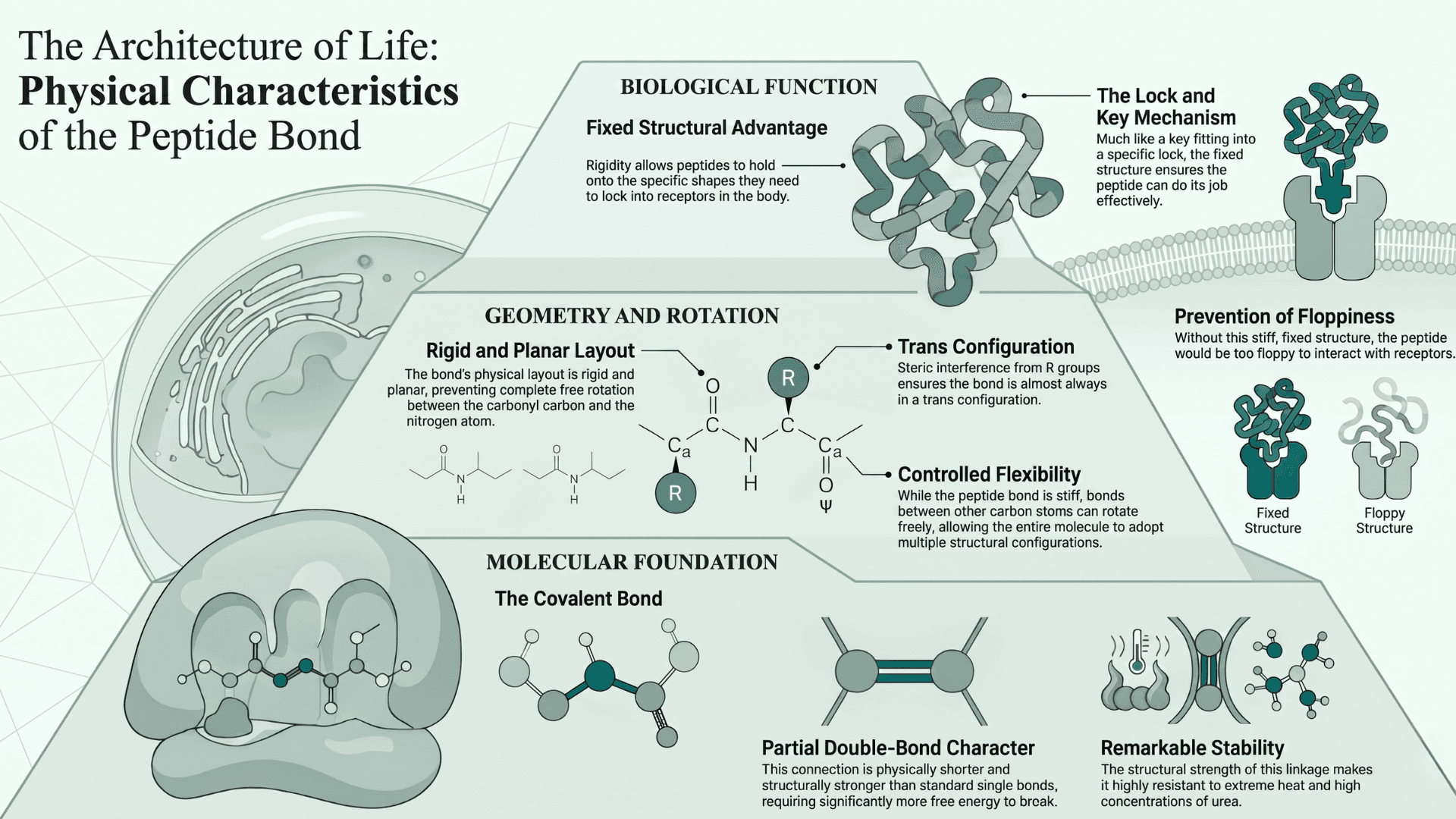
Physical Characteristics of the Bond
Research (Forbes Kaprive et al., NIH) suggests that the resulting peptide bond is remarkably stable. This connection has a partial double-bond character, which makes the bond stiff and prevents the molecules from spinning around freely.
That rigidity is actually a huge advantage. It allows peptides to hold onto the specific shapes they need so they can lock into receptors in the body, much like a key fitting into a specific lock. Without that fixed structure, the peptide would be too floppy to do its job effectively.
The specific connection between two amino acids is called a covalent bond. It forms inside the active site of the ribosome. This connection possesses a partial double bond character. Double bonds are physically shorter and structurally stronger than standard single bonds. They require significantly more free energy to break apart.
This structural strength makes the linkage highly resistant to extreme heat. It also resists high concentrations of urea. As a result, the bond is rigid and planar. Steric interference from R groups ensures it is almost always in a trans configuration.
This physical layout prevents complete free rotation between the carbonyl carbon and the nitrogen atom. However, bonds between other carbon atoms in the sequence can rotate freely. This allows the entire molecule to adopt multiple structural configurations.
How Are Peptides Classified by Chain Length?
The naming depends strictly on the exact number of residues present in the sequence. The chain expands as more units join the primary structure.
A sequence containing between 10 and 20 amino acids receives a specific designation. It is called an oligopeptide. When the sequence exceeds 20 amino acids, it changes categories entirely. It becomes an unbranched chain known as a polypeptide.
Classification Category | Exact Length | Physical Structure |
|---|---|---|
Peptide | 2 to 50 amino acids | Short string |
Oligopeptide | 10 to 20 amino acids | Enlarged chain |
Polypeptide | Greater than 20 amino acids | Unbranched chain |
Short chains often feature specific structural constraints. For example, some nonapeptides share a single disulfide bridge. Long chains form much larger preprohormones. These large structures undergo cleavage within the Golgi apparatus. They break down into smaller prohormones.
The length of the chain is what decides which post-translational changes are necessary. These are essentially chemical tweaks like methylation, phosphorylation, or acetylation that happen after the initial chain is built.
By adding these small groups, the cell can dramatically speed up or slow down how fast it produces these molecules. These adjustments are vital because they turn a raw string of amino acids into a fully functional and high-performance tool.
Important Definitions:
What do we mean by structural constraints?
This refers to physical limitations on how a molecule can move or fold. In short peptides, there is not much room for complex folding. So they often depend on specific chemical staples to hold a functional shape.
What are Nonapeptides?
These are peptides made of exactly nine amino acids.
What is a Disulfide Bridge?
This is a strong covalent bond between two sulfur atoms. In a nonapeptide, this bridge acts like a molecular staple, pulling two parts of the short chain together to create a loop.
This loop shape is often required for the peptide to fit into its specific cellular receptor.
What do we mean by the Maturation Process (Cleavage)?
Longer peptides are often born as much larger. Inactive parent molecules that must be cut down to size. Cleavage is the physical act of cutting the peptide chain. Specialized enzymes act like scissors to snip away the extra amino acids, leaving behind the final, active peptide.
What is a Preprohormone?
This is the initial, large version of a hormone as it is first built by the ribosome. It contains extra segments that act like shipping labels to guide it through the cell.
What is a Prohormone?
Once the shipping label (signal sequence) is removed, the molecule is called a prohormone. It is smaller than the preprohormone but is still usually inactive.
What is the Golgi Apparatus?
It is an organelle that receives proteins and peptides, modifies them, and sorts them for their final destination.
What are Post-Translational Modifications (PTMs)?
These are chemical changes made to the peptide after it has been translated from the genetic code. Adding these chemical tags can turn a peptide on or off. It can also change how long it lasts in the body.
What is a Methylation?
The addition of a methyl group (one carbon and three hydrogen atoms). This often changes how the peptide interacts with other proteins.
What is phosphorylation?
The addition of a phosphoryl group. This is one of the most common ways cells toggle a peptide between an active and inactive state.
What is the definition of Acetylation?
The addition of an acetyl group. This often helps protect the peptide from being broken down too quickly by the body’s enzymes.
Lastly, what do we mean by Synthesis Rates?
This refers to how fast the cell can produce these molecules. The adjustments mentioned (the PTMs) can act as a feedback loop. If a cell sees it has plenty of phosphorylated active peptides, it may slow down the synthesis of new preprohormones to save energy.
How Do Cells create Peptides?
Studies (Forbes Kaprive et al., NIH) explain how these chains are connected and sent to work. In living organisms, the production process begins deep inside the cell nucleus.
An RNA template is transcribed directly from DNA. The messenger RNA travels out to the cytoplasm. It then attaches itself to a ribosome. Once the ribosome is in position, the cell must determine the exact point where the connection begins.
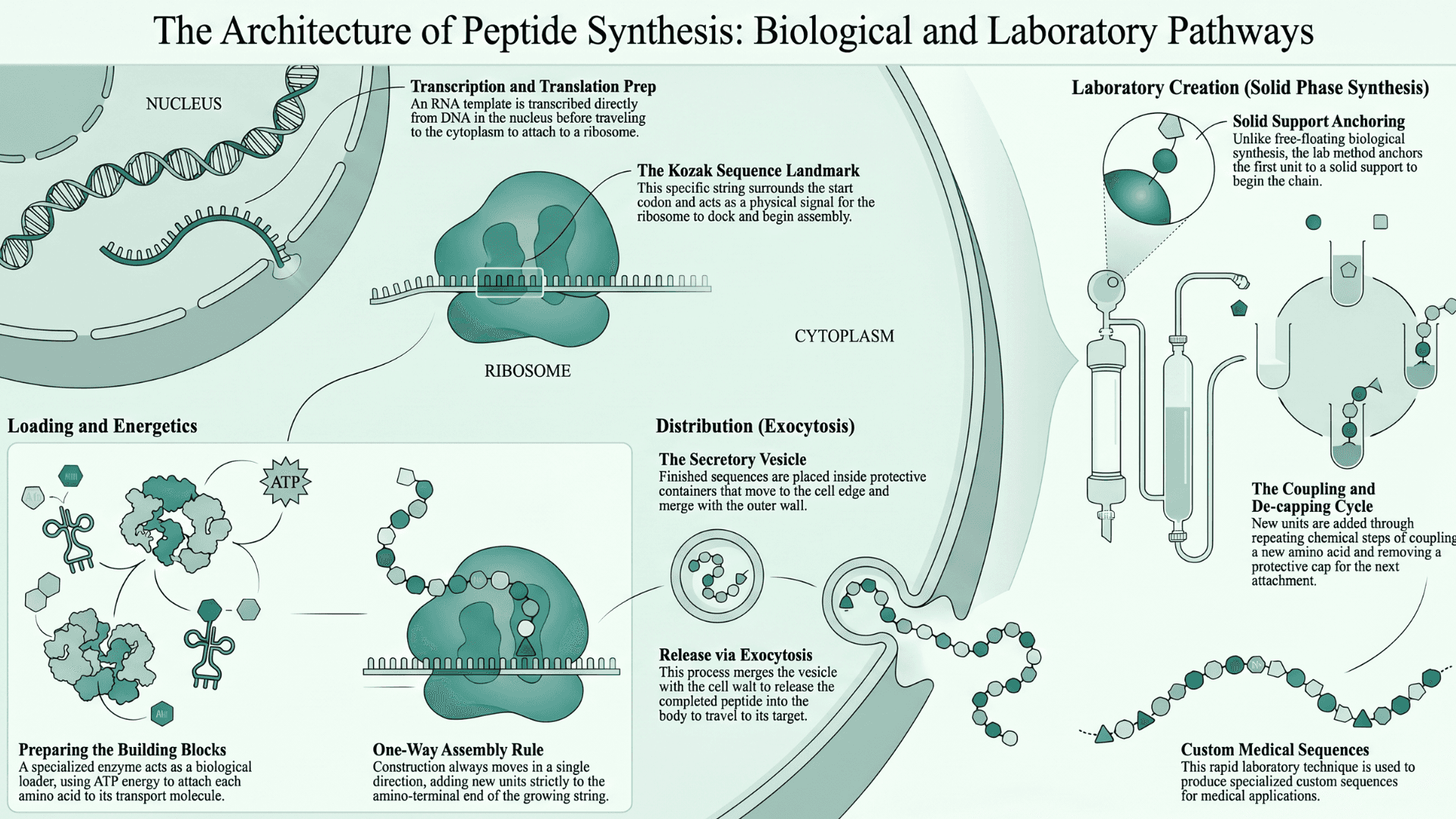
Finding the Start Line
The cell uses a physical landmark to know exactly where to begin building. This landmark is a specific string called a Kozak sequence. It surrounds the start codon and acts as a signal for the ribosome to dock and start the assembly.
Loading the Building Blocks
Each amino acid must be prepared before it can join the chain. This preparation requires cellular energy called ATP. A specialized enzyme acts as a biological loader to attach each amino acid to a transport molecule. This enzyme ensures the correct building block is ready for the assembly line.
One Way Assembly
The construction always moves in a single direction. Every new unit is added only to the amino-terminal end of the growing string. This strict rule ensures the sequence matches the genetic code exactly.
Shipping the Final Product
Once the sequence is finished, it is placed inside a protective container called a secretory vesicle. This container moves to the edge of the cell and merges with the outer wall. This process is called exocytosis. It releases the completed peptide into the body so it can travel to its target.
Laboratory Creation
Scientists build these molecules differently in a lab using a method called solid phase synthesis. They anchor the first unit to a solid support and then add new units through repeating chemical steps.
These steps involve coupling the new amino acid and then removing a protective cap so the next one can attach. This laboratory technique allows for the rapid production of custom sequences for medical use.
What Roles Do Peptides Play in the Human Body?
While research is still ongoing, current data (Kearly et al., NIH) shows their critical functions in growth, development, and stress responses.
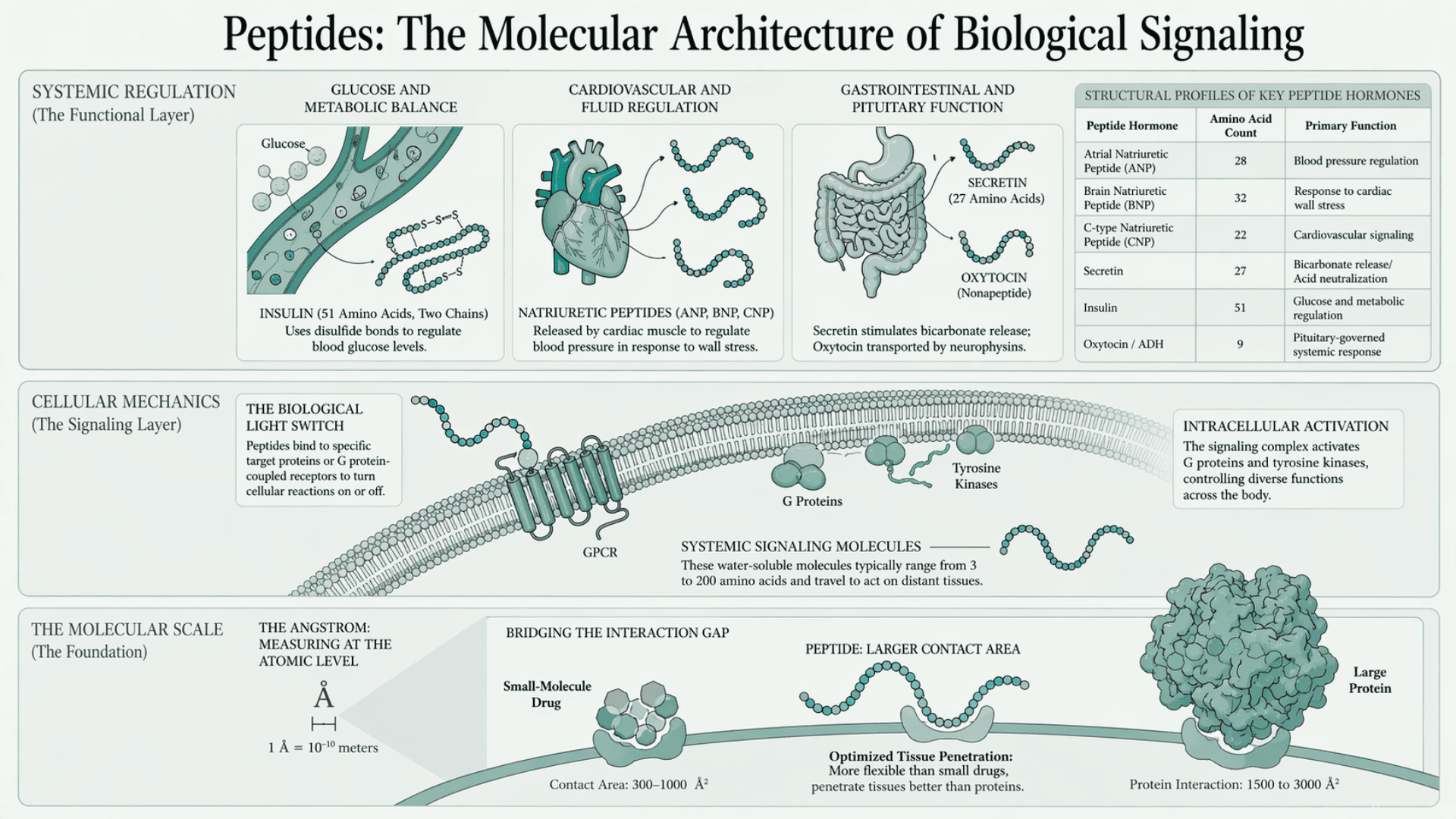
They function as strong signaling molecules. They bind to specific target proteins or G protein-coupled receptors. This binding forms a complex that acts like a biological light switch. It turns cellular reactions on or off. The signal then activates other proteins, including G proteins and tyrosine kinases. This process controls many functions across the body.
There is evidence (Han et al., NIH) that these structures regulate complex immune systems and metabolic pathways. Small molecules bridge the gap between high specificity and manufacturing costs. Small-molecule drugs have a very limited contact area. This area measures between 300 and 1000 square angstroms.
An angstrom is a very small unit used to measure distances at the atomic level. It equals one ten-billionth of a meter. In chemistry, it is commonly used to describe the distance between atoms or the length of chemical bonds. For example, a typical carbon–hydrogen bond is about 1 angstrom long.
Mathematically, it is written as:
1 Å = 10⁻¹⁰ meters
Most atoms are about 1 to 2 angstroms in size. Because of this, the angstrom is a practical unit for working at the molecular scale, instead of using long decimal values in meters or nanometers.
Protein interactions cover a much larger area of 1500 to 3000 square angstroms. These short chains are larger than small drugs. This allows them to be more flexible when binding to receptors. They also penetrate tissues more effectively than large proteins.
Peptide Hormones and Systemic Regulation
Hormonal activity is a major part of how peptides function in the body. These water-soluble molecules typically range from 3 to 200 amino acids in length. They are produced in one location but can travel to act on distant tissues.
Their primary role is to regulate growth and cellular differentiation. Some peptides control essential systemic functions. For example, insulin is a peptide made of 51 amino acids. It consists of two chains linked by disulfide bonds. It regulates glucose levels and maintains metabolic balance.
Oxytocin and antidiuretic hormone are produced in the posterior pituitary gland. Both are nonapeptides with a single disulfide bond. They differ by only two amino acids. Carrier proteins called neurophysins transport these peptides within the body.
Secretin is another peptide hormone. It contains 27 amino acids. It stimulates the pancreas and bile ducts to release bicarbonate. This helps neutralize stomach acid.
Natriuretic peptides are released by cardiac muscle cells in response to wall stress. They help regulate blood pressure.
Atrial natriuretic peptide contains 28 amino acids. Brain natriuretic peptide contains 32 amino acids. C-type natriuretic peptide contains 22 amino acids. These peptides share structural similarities.
The diversity of these peptide hormones allows precise control over metabolism and cardiovascular function. As more research is being done, we are uncovering new frontiers into how beneficial peptides are for human beings.
It should come as no surprise that beyond natural biology, peptides are now widely used in medicine and drug development.
What Role Do Peptides Play in Advanced Therapies and Drug Development?
The evidence (Xiao et al., Nature) is there that Pharmaceutical companies are heavily investing in peptides. They have started to combine these short sequences with other chemical payloads.
These combinations create highly targeted delivery systems called conjugates. These conjugates transport drugs directly to diseased cells. They spare healthy tissue from collateral damage. This targeted approach is completely changing cancer treatment, for example.
The clinical trial ecosystem is massive already. Hundreds of new variations are currently in Phase 1, Phase 2, and Phase 3 clinical trials. They target a wide range of severe human conditions.
The main focus areas include metabolic diseases, cardiovascular conditions, and cancer. Researchers are working on ways to make these drugs last longer in the body. One key concept is peptide half-life. It is the time it takes for half of a drug to break down or be removed from the body.
So, how are scientists extending the half-life of peptides?
By using techniques like PEGylation and lipidation:
PEG stands for polyethylene glycol. It is a synthetic, water-soluble compound made from repeating units of ethylene glycol. In simple terms, it is a long, flexible chain that mixes easily with water. It is used in medicine to improve stability and help drugs stay in the body longer.
PEGylation means attaching a polyethylene glycol (PEG) chain to the peptide. This added chain makes the molecule larger and shields it from enzymes that would normally break it down. It also reduces how quickly the body clears it from the bloodstream.
Lipidation means attaching a lipid (fat molecule) to the peptide. This helps the peptide bind to proteins in the blood, such as albumin. Because of this binding, the peptide stays in circulation longer and is released more slowly over time.
These modifications help protect the drug from being broken down too quickly. As a result, the drug stays in the bloodstream for longer and reduces the need for frequent injections.
How Are Peptides Used to Deliver Drugs into Cells?
Getting medication inside a cell is a major biological challenge. Cell membranes act as protective barriers that prevent most substances from entering. Certain short sequences have the ability to cross this barrier. These are known as cell-penetrating peptides.
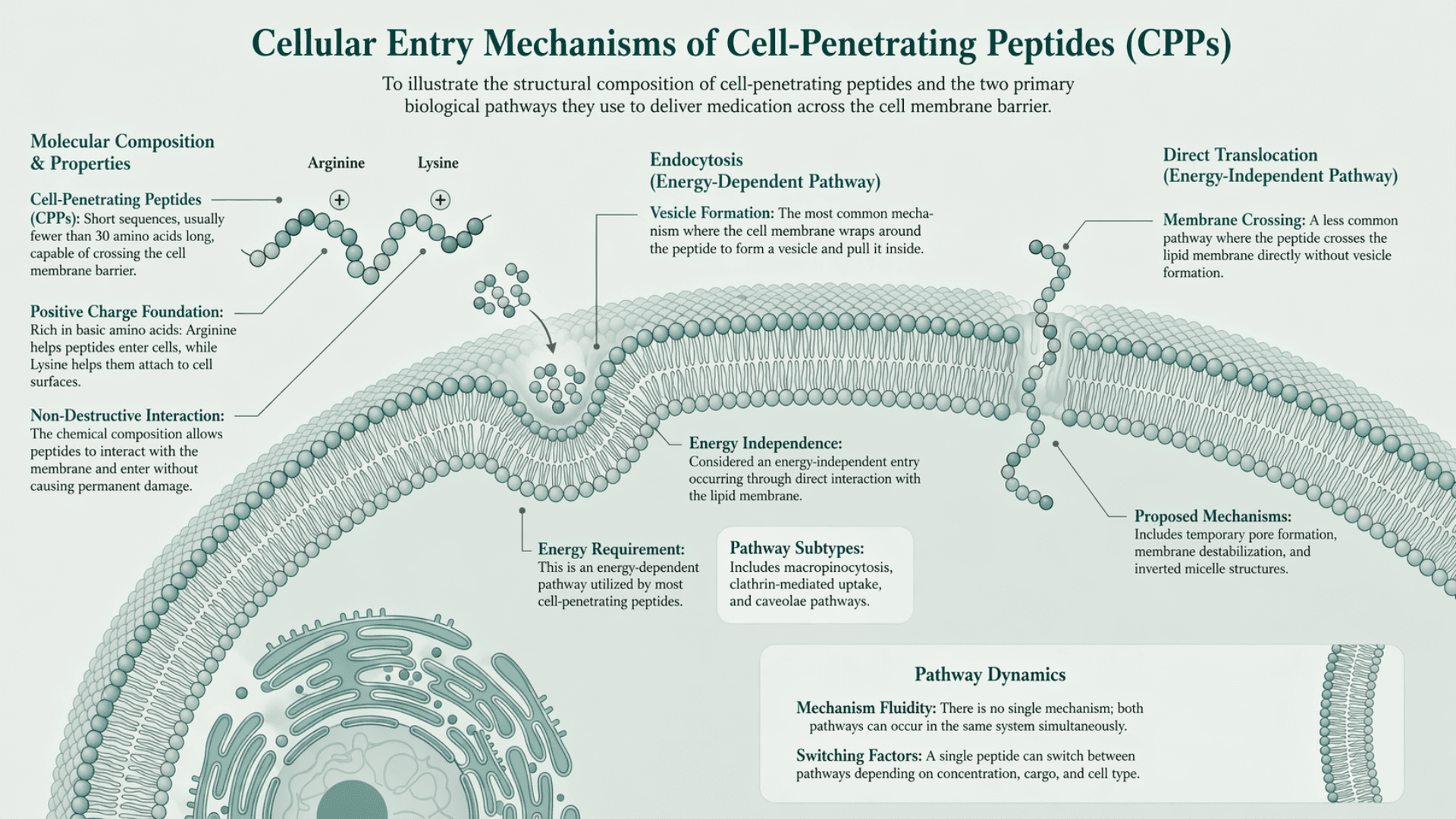
They are usually fewer than 30 amino acids long and are rich in basic amino acids such as arginine and lysine. Arginine is a positively charged amino acid that helps peptides enter cells. Lysine is a positively charged amino acid that helps peptides attach to cell surfaces and get inside. This chemical composition allows them to interact with the cell membrane and enter without causing permanent damage.
There is no single mechanism for how cell-penetrating peptides enter cells. Research consistently shows two main pathways:
Endocytosis is the most common mechanism. In this process, the cell membrane wraps around the peptide, forms a vesicle, and pulls it inside the cell. This is an energy-dependent pathway used by most cell-penetrating peptides (Lee et al., Nature).
There are multiple subtypes, including macropinocytosis, clathrin-mediated uptake, and caveolae pathways. All of these fall under endocytosis.
Direct translocation is less common and more debated. In this pathway, the peptide crosses the membrane directly without vesicle formation or classical uptake machinery. It occurs through interaction with the lipid membrane and is considered an energy-independent entry (Hernandez & Marin, Springer)
Proposed mechanisms include temporary pore formation, membrane destabilization, and inverted micelle structures (Trabulo et al., NIH).
Both mechanisms can occur in the same system. The same peptide can switch between pathways depending on concentration, cargo, and cell type (Ruseska & Zimmer, NIH).
How Do Antimicrobial Peptides Kill Pathogens?
A recent study (Zheng et al., NIH) explains exactly how peptides fight off dangerous infections. They form a critical part of the human immune system. The epithelium, neutrophils, mast cells, and adipocytes produce them naturally. They serve as a very rapid first line of biological defense.
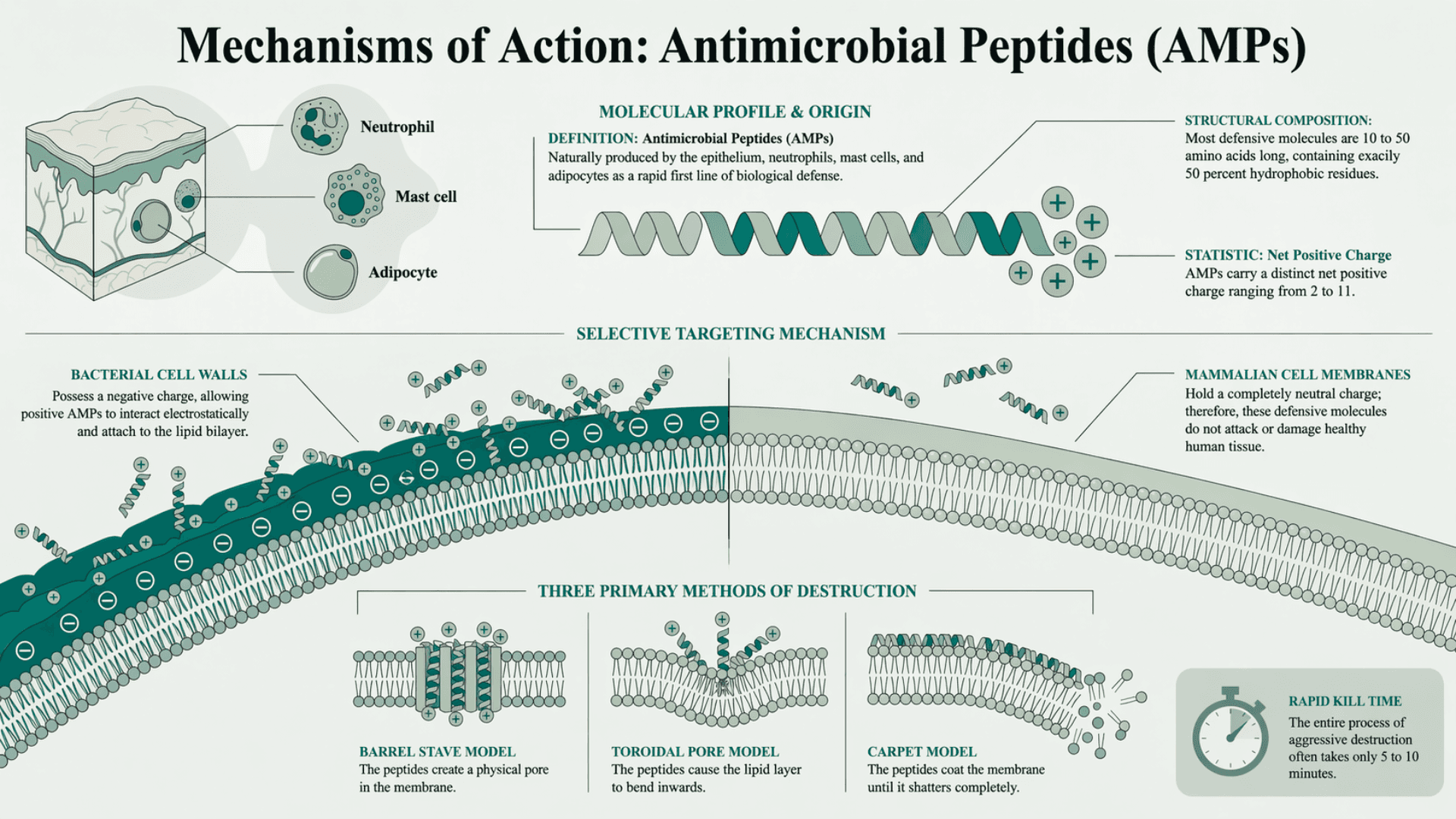
Most of these defensive molecules are 10 to 50 amino acids long. They carry a distinct net positive charge. This charge typically ranges from 2 to 11. They also contain a very large percentage of hydrophobic residues. These residues usually make up exactly 50 percent of the total structure.
This specific physical makeup is highly weaponized. The positive charge allows them to interact electrostatically with invading bacteria. Bacterial cell walls possess a negative charge. The molecules attach to the bacteria and aggressively destroy the lipid bilayer.
They execute this destruction through three primary methods:
The barrel stave model creates a physical pore in the membrane.
The toroidal pore model bends the lipid layer inwards.
The carpet model coats the membrane until it shatters completely.
This allows them to kill invading microorganisms extremely fast. The entire process often takes only 5 to 10 minutes. Mammalian cell membranes hold a completely neutral charge. Therefore, these defensive molecules do not attack or damage healthy human tissue.
How Do Peptides Control Plant Growth and Stress Response?
But it's not just human beings who are benefiting from peptides. Increasing evidence (Kearly et al., OUP) has been uncovered that shows how these molecules change plant biology. They function as strong signaling transmitters within agricultural systems.
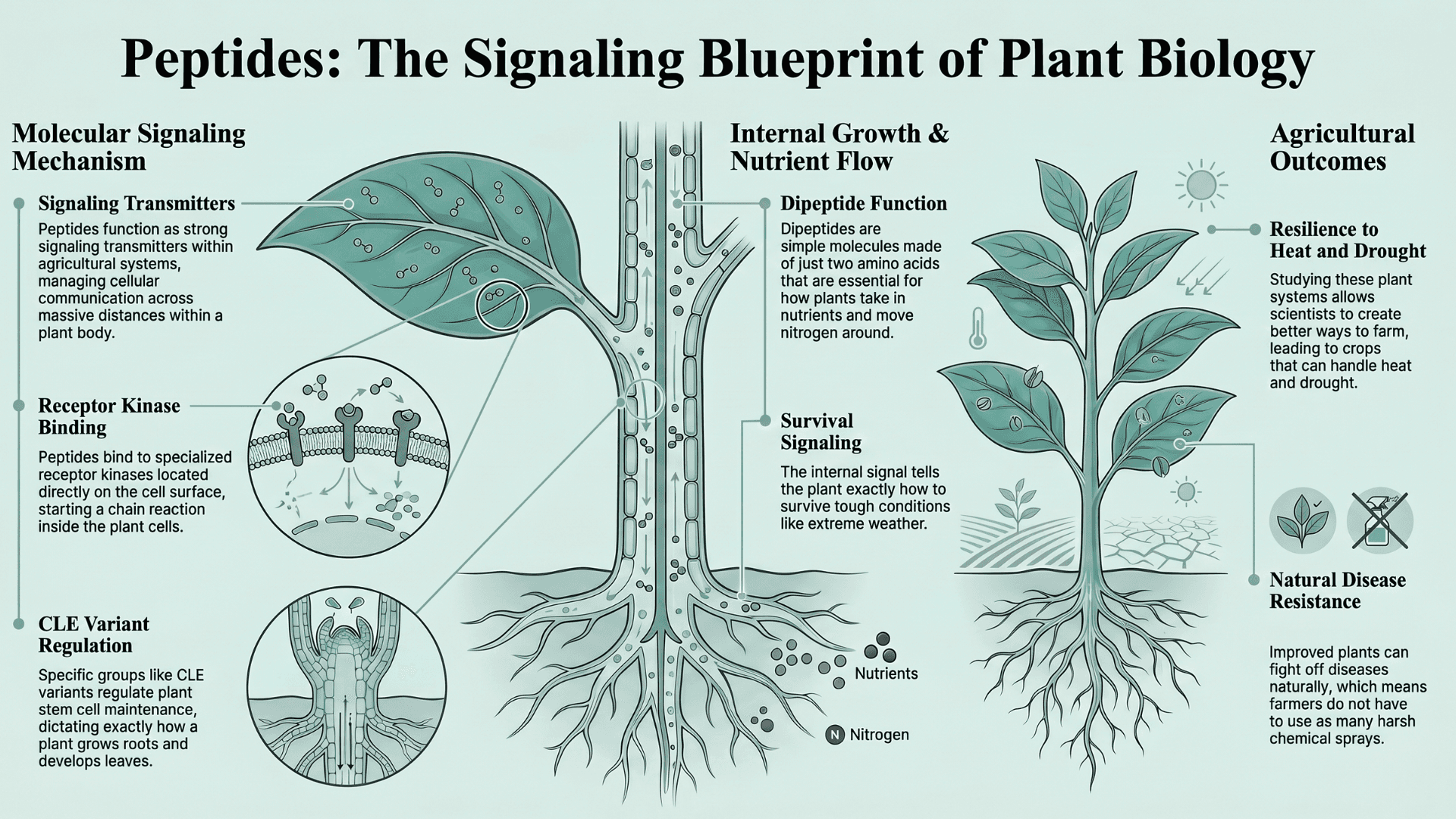
They manage cellular communication across massive distances within a plant body. Specific groups like CLE variants regulate plant stem cell maintenance. They dictate exactly how a plant grows roots and develops leaves. They bind to specialized receptor kinases located directly on the cell surface.
When this binds, it starts a chain reaction inside the plant cells. This signal tells the plant exactly how to survive tough conditions like extreme weather. Dipeptides are simple molecules made of just two amino acids. Even though they are small, they are essential for how plants take in nutrients and move nitrogen around.
By studying how these plant systems work, scientists can create better ways to farm. This leads to crops that can handle heat and drought. These improved plants can also fight off diseases naturally, which means farmers do not have to use as many harsh chemical sprays.
How Are Peptides Extracted and Used from Industrial Byproducts?
With the peptide market growing at an unprecedented rate, studies (Colón-Sandoval et al., NIH) highlight unexplored opportunities in the modern circular economy.
The global food industry generates a huge amount of industrial waste every year. Scientists are now extracting valuable sequences directly from this waste material. They utilize animal, plant, and marine byproducts. This extraction process creates high-value nutraceuticals from discarded industrial garbage.
Vegetable byproduct derivatives are excellent sources for cardiometabolic functional foods. Pumpkin oil cake and black sesame cake yield specific compounds. These compounds naturally lower high blood pressure. They actively inhibit the angiotensin converting enzyme inside the human body.
Marine byproducts generate unique molecules with powerful antibacterial properties. They also provide potent anti-inflammatory health benefits. These marine extracts show massive potential for developing new pharmacological agents. They offer a sustainable way to manage chronic inflammation and fight emerging pathogens.
However, major industrial challenges still exist today. Translating laboratory success into commercial products is very difficult and expensive. Manufacturers must address the choice of administration systems for consumers.
Companies really need reliable ways to make these products the same way every time. The methods used to break down proteins with enzymes must be identical in every factory. Cleaning and filtering steps have to follow strict international rules.
Future research needs to look closely at both the technology and the costs. Turning leftover protein waste into something valuable only works if it stays cheaper than using man-made chemicals.
The Peptide Revolution, the next Cultural Phenomenon
This is not a sudden out-of-the-blue event. Every data (Brookshire, Scientific American) shows that the interest in peptides is escalating rapidly. And it isn’t just social media hype or trend. There is scientific evidence.
The success of semaglutide drugs like Ozempic and Wegovy changed how the world views metabolic treatment. As these options proved effective, the stigma around injections faded. People became comfortable with subcutaneous injections at home.
The cultural tipping point came around 2022. More and more people sought out unapproved compounds. They searched for molecules like BPC 157 and TB 500 online. Many of them had reasons like the inability to find health solutions through conventional channels. But with that came more awareness by regulatory bodies to safeguard the health of consumers.
What are the Global Rules related to Peptides?
Peptides are regulated by different organizations depending on their use in medicine or sports. These rules vary by country but follow similar safety goals.
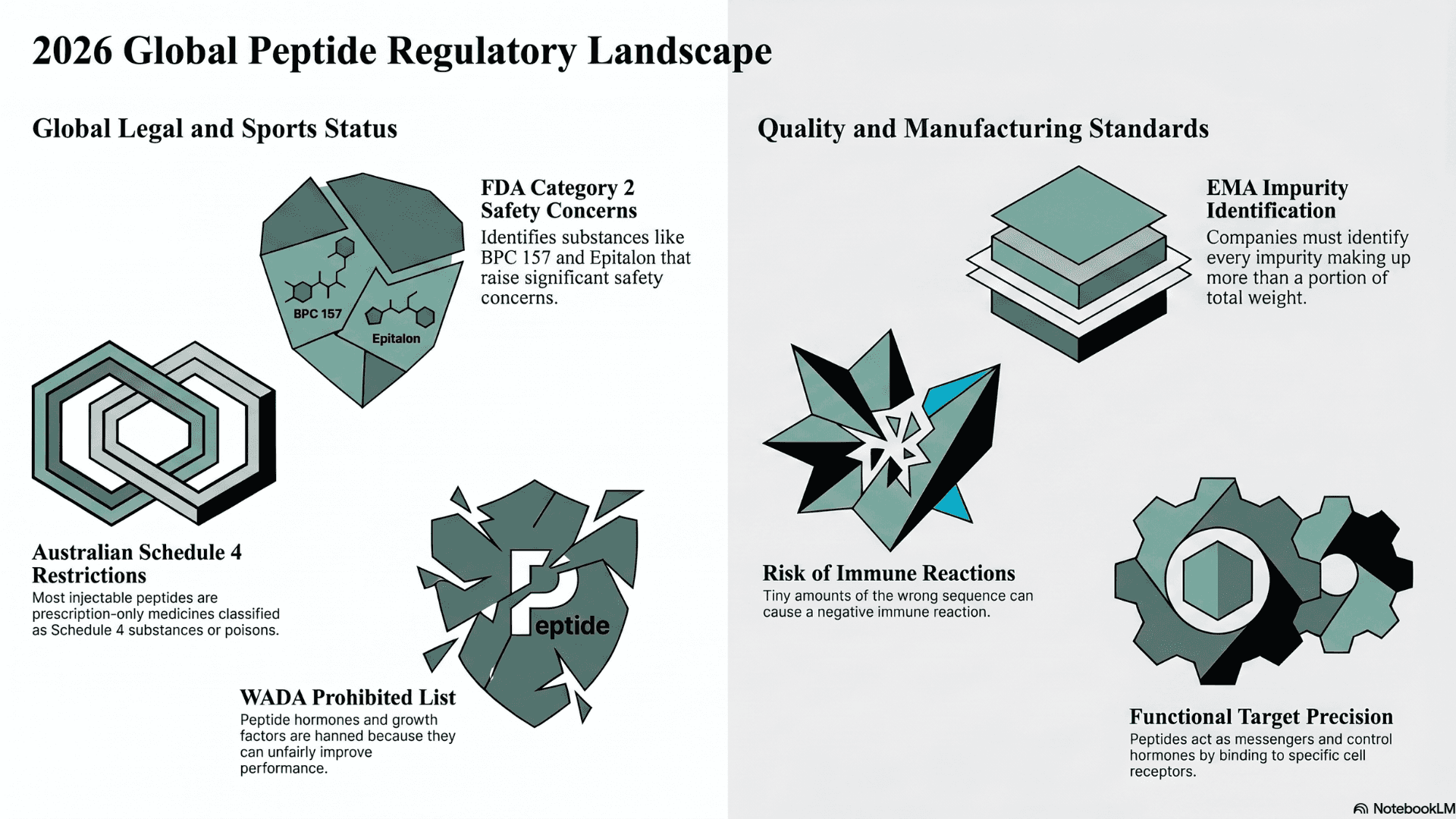
United States Compounding Status
An official document (FDA) released in April 2026 lists many peptides under category 2. This category identifies substances that raise significant safety concerns. The list includes popular options like BPC 157 and Epitalon, along with Ibutamoren Mesylate and Melanotan II.
Another federal notice (GPO) provides more detail on upcoming changes. A specialized committee will meet on July 23 and 24, 2026. They will discuss moving several substances to a list that allows for safe compounding. The meeting will focus on BPC 157 and KPV as well as TB 500 and MOTS-c.
This review process is ongoing. Until the committee makes a final decision, these substances remain restricted. The agency continues to warn that many unapproved chains lack sufficient safety data for human use.
International Sports Rules
The World Anti-Doping Agency sets the standards for professional sports worldwide. According to the 2026 prohibited list (WADA), many peptides are banned at all times. This includes peptide hormones and growth factors under the S2 category. These substances are prohibited because they can unfairly improve performance. They also present potential health risks to athletes.
The 2026 update includes specific examples of banned substances like Pegmolesatide. The agency is also monitoring the use of weight loss markers like semaglutide. This international code applies to every organization that signs the agreement. It means a substance banned by this agency is restricted in almost every professional sport globally.
Australian Medical Restrictions
In Australia, the Therapeutic Goods Administration manages the legal status of these compounds. A safety alert (TGA) confirms that most injectable peptides are prescription-only medicines. They are classified as Schedule 4 substances.
It is illegal to possess or sell these items in Australia without a valid script from a doctor. This includes specific molecules like BPC 157 and TB 500. The regulator classifies these as restricted substances or poisons. Selling them without authorization can result in heavy fines or jail time. Promoting these unapproved products on social media is also a breach of national law.
European Manufacturing Standards
An official scientific guideline (EMA Europa) from the European Medicines Agency sets strict quality rules. This document outlines exactly how companies must manufacture synthetic chains. They must identify every single impurity that makes up more than a portion of the total weight.
These rules exist because even tiny amounts of the wrong sequence can be dangerous. Impurities can cause the human body to have a negative immune reaction. Therefore, companies must use very precise testing methods.
This level of testing ensures that every medicine is safe and consistent. It makes the approval process for new therapies slow and expensive. However, it protects patients from the risks of low-quality or contaminated products.
So, peptides are short chains that link single amino acids to large proteins. Their length and sequence allow them to perform exact tasks in the body. They act as messengers and control hormones by binding to cell receptors. This ability to pick a target makes them vital for life and for new medicine. By learning how they form and how they are grouped, we find the path to better drugs and new ways to heal.
FAQ
How are therapeutic peptides tested for receptor binding?
Using lab methods like SPR, ITC, and ELISA to measure binding strength and specificity.
What is the structure–activity relationship in peptide design?
It explains how small changes in sequence or structure affect biological activity.
Why do some peptides trigger immune responses?
The body may recognize modified or synthetic sequences as foreign and activate immunity.
What is peptide immunogenicity?
It is the potential of a peptide to cause an immune reaction in the body.
How do peptides cross biological barriers like the blood–brain barrier?
Only specific modified or small peptides can pass through, often using transport systems.
What is the difference between peptide drugs and biologics?
Peptides are chemically synthesized, while biologics are produced from living systems.
Why are peptide drugs expensive to develop?
They require complex synthesis, purification, and strict regulatory testing.
What is peptide degradation in the body?
Enzymes rapidly break peptides into smaller fragments, reducing their effectiveness.
What is the role of peptide sequencing in drug development?
It determines the exact amino acid order needed for correct biological function.
. Can therapeutic peptides be personalized?
Yes. Sequences can be designed or adjusted to target specific pathways or patients.



